![PDF] Effect of Preoperative Ingestion of Orange Juice on pH and Volume of Gastric Contents | Semantic Scholar PDF] Effect of Preoperative Ingestion of Orange Juice on pH and Volume of Gastric Contents | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6b916b8e673a54a8d4539385bf07ee20a0b65ff4/2-Table1-1.png)
PDF] Effect of Preoperative Ingestion of Orange Juice on pH and Volume of Gastric Contents | Semantic Scholar

SciELO - Brasil - In vitro evaluation of the effect of natural orange juices on dentin morphology In vitro evaluation of the effect of natural orange juices on dentin morphology

A comparative study of pH modulation and trace elements of various fruit juices on enamel erosion: An in vitro study Nirmala S, Subba Reddy V V - J Indian Soc Pedod Prev Dent
![SOLVED:Use the following definition. In chemistry, the pH of a solution is defined to be pH=-log[H^+], where H^+ is the hydrogen ion concentration of the solution in moles per liter. Distilled water SOLVED:Use the following definition. In chemistry, the pH of a solution is defined to be pH=-log[H^+], where H^+ is the hydrogen ion concentration of the solution in moles per liter. Distilled water](https://cdn.numerade.com/previews/fb6e396d-397f-464b-9056-d248e444218c_large.jpg)
SOLVED:Use the following definition. In chemistry, the pH of a solution is defined to be pH=-log[H^+], where H^+ is the hydrogen ion concentration of the solution in moles per liter. Distilled water

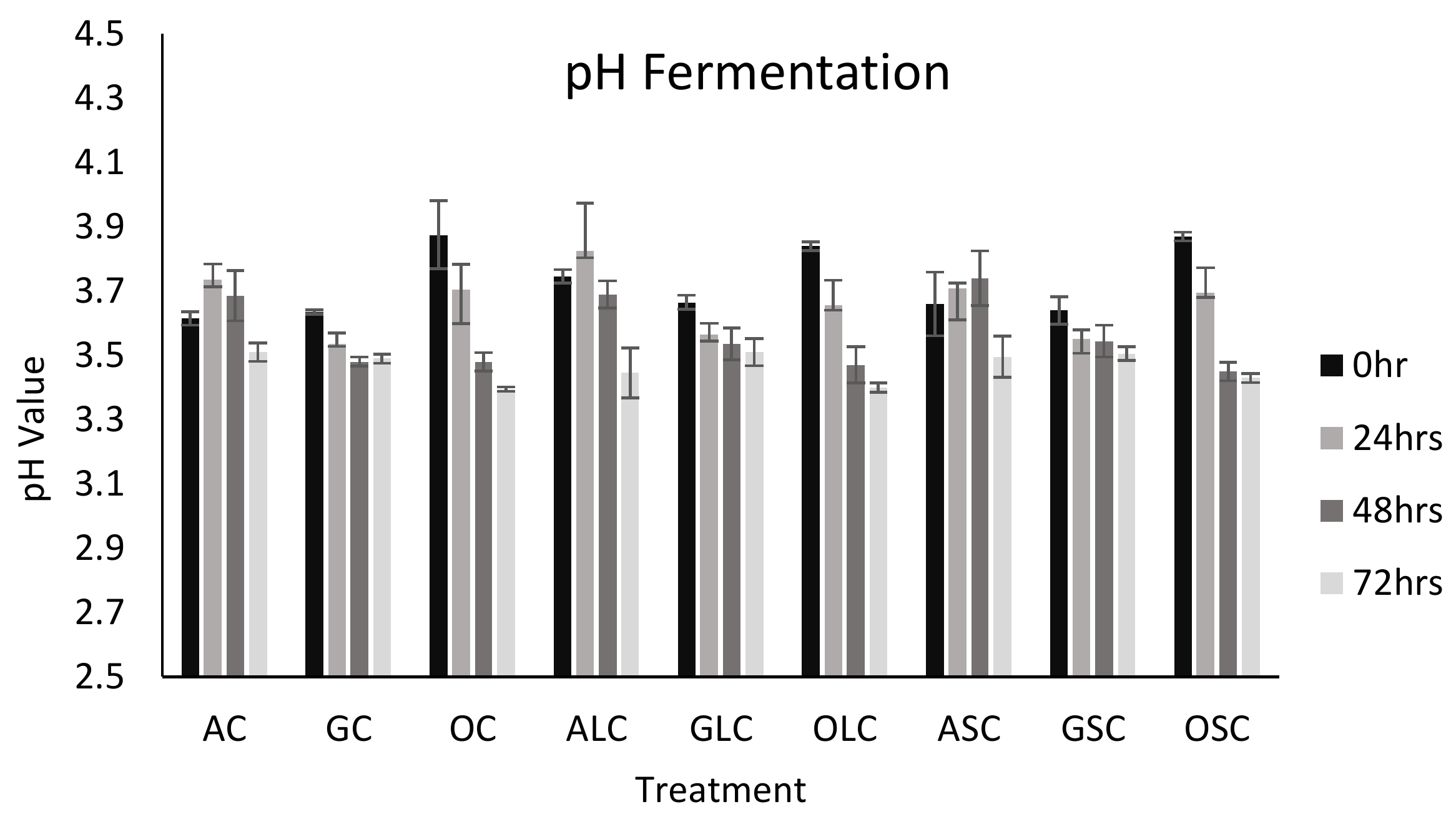
Fermented Orange Juice: Source of Higher Carotenoid and Flavanone Contents | Journal of Agricultural and Food Chemistry